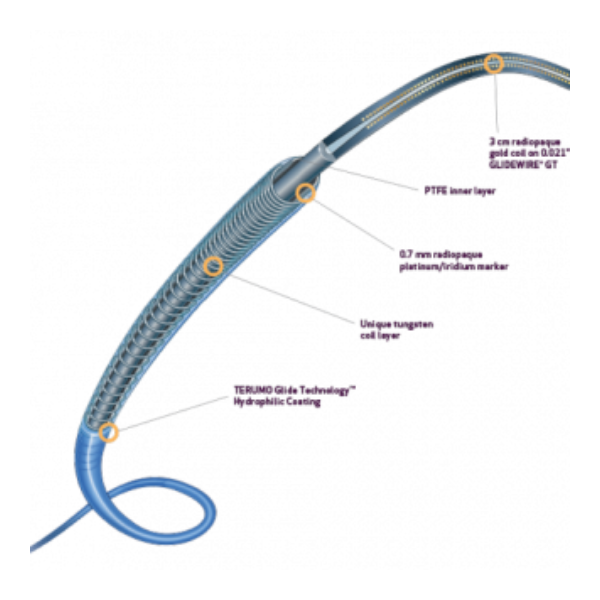
| Fr Size | Product Code | Length (cm) | Max Pressure (PSI) | RO Markers | Hydrophilic Coating Length (cm) | Glidewire® GT Length (cm) | Glidewire® GT Size (in) |
|---|---|---|---|---|---|---|---|
| 2.4 Fr | MC*PE24111YB | 110 | 750 | 1 | 50 | 120 | 0.018 |
| 2.4 Fr | MC*PE24131YB | 130 | 750 | 1 | 70 | 140 | 0.018 |
| 2.4 Fr | MC*PE24151YV | 150 | 750 | 2 | 90 | 160 | 0.018 |
| 2.7 Fr | MC*PE27111Y | 110 | 750 | – | 50 | 120 | 0.021 |
| 2.7 Fr | MC*PE27131Y | 130 | 750 | – | 70 | 140 | 0.021 |
| 2.8 Fr | MC*PE28111YB | 110 | 900 | 1 | 50 | 120 | 0.021 |
| 2.8 Fr | MC*PE28131YB | 130 | 900 | 1 | 70 | 140 | 0.021 |
| 2.8 Fr | MC*PE28151YV | 150 | 900 | 2 | 90 | 160 | 0.021 |
PRODUCT SPECIFICATIONS
| Catheter OD | Length (cm) | Inner Diameter (in/mm) | Max GW (in) | Embolic Compatibility | Dead Space Volume (mL) | Actual Flow Rate* (mL/sec) @ 750 psi | Actual Flow Rate* (mL/sec) @ 900 psi |
|---|---|---|---|---|---|---|---|
| 2.4/2.9Fr (0.80/0.97mm) | 110 | 0.022″/0.57 | 0.018″ | 0.018″ Coils/HydroPearl® 600 ± 75 μm | 0.48 | 2.3 | – |
| 2.4/2.9Fr (0.80/0.97mm) | 130 | 0.022″/0.57 | 0.018″ | 0.018″ Coils/HydroPearl® 600 ± 75 μm | 0.53 | 2 | – |
| 2.4/2.9Fr (0.80/0.97mm) | 150 | 0.022″/0.57 | 0.018″ | 0.018″ Coils/HydroPearl® 600 ± 75 μm | 0.58 | 1.8 | – |
| 2.7/2.9Fr (0.90/0.97mm) | 110 | 0.025″/0.065 | 0.021″ | 0.018″ Coils/HydroPearl® 800 ± 75 μm | 0.57 | 3.5 | – |
| 2.7/2.9Fr (0.90/0.97mm) | 130 | 0.025″/0.065 | 0.021″ | 0.018″ Coils/HydroPearl® 800 ± 75 μm | 0.64 | 3.2 | – |
| 2.8/3.0Fr (0.93/1.00mm) | 110 | 0.027″/0.70 | 0.021″ | 0.018″ Coils/HydroPearl® 800 ± 75 μm | 0.61 | 4.1 | 4.7 |
| 2.8/3.0Fr (0.93/1.00mm) | 130 | 0.027″/0.70 | 0.021″ | 0.018″ Coils/HydroPearl® 800 ± 75 μm | 0.68 | 3.7 | 4.3 |
| 2.8/3.0Fr (0.93/1.00mm) | 150 | 0.027″/0.70 | 0.021″ | 0.018″ Coils/HydroPearl® 800 ± 75 μm | 0.75 | 3.1 | 3.9 |
REFERENCES
RX ONLY. Refer to the product labels and package insert for complete warnings, precautions, potential complications, and instructions for use.
- Catheter [Instructions for Use]. Somerset, NJ: Terumo Medical Corporation; 2016
- PROGREAT Catheter 510(k); 2003
- PROGREAT DMSO Compatibility Statement Letter
- Data on file.